Welcome to the Kandel Group Image Gallery.
There are currently 82770 data sets, acquired from November 2001 to January 2024, available to view.
Recent Uploads:
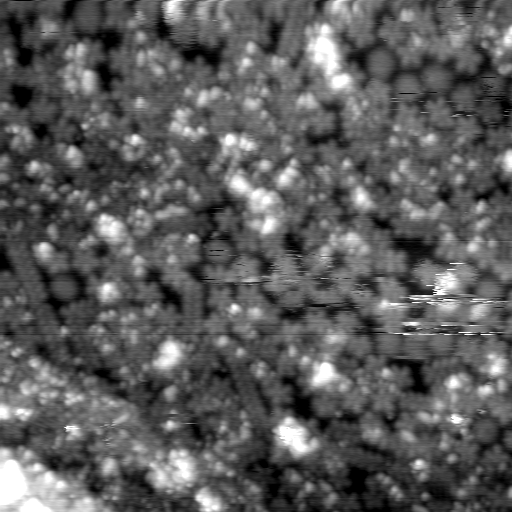
- 2x1000us 20mM melamine in water/methanol 75/25 on Au(111) single crystal, possible miss on pulse: 10 data sets acquired on Jan. 26, 2024.
You must log in to upload data or enter comments.
The Group:
 |
 |
| David Turner, Joe Coman, Angela Silski, Jacob Petersen, Catlin Schalk, Ryan Brown, Ashley Gans, and Alex Kandel | Photomosaic using STM images from gallery (click to enlarge) |
The Kandel group is in the Department of Chemistry and Biochemistry in the College of Science at the University of Notre Dame
Prof. Kandel's faculty webpage and CV
The Research:
Scanning Tunneling Microscopy (STM), with its ability to acquire atomic-scale images of solid surfaces, is our primary tool for investigating the physical properties and chemical reactivity of these surfaces:
- The importance of local surface environment and structure on chemical reactivity is measured using sequences of STM images acquired in situ during gas-surface chemical reactions. We acquire "stop motion" movies to study the effect of hydrogen-atom (JACS, JPC C, JPC C) and chlorine-atom (JCP, PCCP, JCP, JPC Letters) reaction with alkanethiol self-assembled monolayers.
- The structural and electronic properties of large, organometallic molecules will determine their suitability as components for molecular electronic devices. We record STM images of mixed-valence compounds and have shown that charge can be localized on a single metal center within a larger molecule.(JACS, JACS, JPC C, PCCP)
- The structure of self-assembled monolayers depends on both the molecule-molecule and molecule-surface interaction strengths, as well as on the kinetics of monolayer formation. We have studied monolayers and mixed monolayers of a variety of molecular species on metal surfaces, including fullerenes (Langmuir, Surface Science) and alkanethiols.(Langmuir, JPC C)
- Hydrogen bonds are widely used in supramolecular and surface chemistry to prepare complex structures through self-assembly. We investigate particularly complex assemblies that result from rapid solvent evaporation.(Nature, Chem. Communications)
- We develop and build hardware and electronics. Source files and build instructions can be found on our Open Hardware site.
The Kandel group currently has positions open for interested graduate students. Click here to apply for graduate school at the University of Notre Dame.